Introduction
Frozen embryo transfer (FET) has become the standard of care in assisted reproductive technology practices globally. Different cycle regimens available for endometrial preparation for a FET cycle are artificial cycle (AC), natural cycle-true (tNC) and modified (mNC) and a stimulated cycle.1,2 Consistent evidence from observational studies and a recent truncated randomised controlled trial, emphasizes the increased obstetric and perinatal risks, specifically hypertensive disorders of pregnancy (HDP), preeclampsia and placental bed disorders in AC-FET pregnancies compared to those by NC-FET.3,4 The suboptimal endometrial preparation and absence of ovulation/corpus luteal vasoactive peptides mainly relaxin-2 has been implicated as the cause(s) of these adverse outcomes.5
In this context, the use of ovulatory FET protocols like tNC and mNC are recommended in ovulatory patients.3,6 Challenging issues with tNC-FET protocol is tracking ovulation, frequent visits and scheduling.7 Adding human chorionic gonadotropin (hCG) trigger to the mNC-FET cycle reduces the monitoring and adds extra flexibility of 1–2 days.8 Significance of the association of luteinizing hormone (LH) surge and the downstream effect of the hCG trigger on the endocrine parameters and endometrium receptivity are not clearly defined.9,10 A study reported improved flexibility of 5–7 days by triggering the follicle from size 13–22 mm.11 Triggering at smaller follicle diameters may theoretically not have a functional or a fully functional corpus luteum (CL).12 Lately a new protocol has been studied and addressed by different study groups as a modification of the modified natural protocol,13 natural proliferative phase (NPP),14–16 progesterone–modified natural cycle,17 and programmed ovulatory FET protocol.18 This protocol is elaborated by Godinho et al., in the natural cycle when endometrial thickness (EMT) is ≥ 7 mm and serum progesterone is < 1.5 ng/ml then vaginal micronized progesterone 400 mg every 12 hours is started regardless of the dominant follicle diameter and circulating serum LH levels.14 Blastocyst transfer is scheduled on the 6th day of exogenous progesterone exposure. A recent systematic review and meta-analysis reported that the NPP-FET protocol offers enhanced scheduling convenience without compromising reproductive outcomes, but high-quality trials are still warranted.19 Though it is simple to monitor, flexible and assured progesterone exposure from luteal day one, there is no confirmation regarding a functional CL.12 Further, it has been questioned in literature whether the CL formed from smaller follicles in NPP-FET protocol has functional activity comparable to that of a CL in a natural cycle, particularly with respect to the production of relaxin and other vasoactive substances required to exert the same protective effects during pregnancy.12 It is presumed by the study group that the CL might be induced by either the initiated higher dose of exogenous progesterone or by the non-opposition of the natural follicular growth and luteinization.14 We need evidence for ovulation and CL activity so as to ensure favorable pregnancy outcomes in this protocol. In the NPP-FET protocol as exogenous progesterone is started prior to ovulation unrelated to the preovulatory estradiol rise, this may have negative feedback on the estradiol induced LH surge.20,21 Evidence of ovulation in the NPP-FET protocol has been assessed in two studies, and in both dydrogesterone was administered as luteal phase support (LPS).16,18
Dydrogesterone as such may not inhibit ovulation as reported in some studies.22–24 The more widely used progesterone for LPS in FET cycles is natural progesterone which may have a different endocrine effect on ovulation compared to dydrogesterone. We proposed to study the evidence of ovulation and CL activity in NPP-FET cycles with day 3/day 5 embryo transfer when natural progesterone (intramuscular and vaginal gel) is used as LPS by estimating serum relaxin-2 levels in early pregnancy. Circulating relaxin-2 being solely secreted by the CL in pregnancy can be taken as an index of CL activity and an indirect evidence of ovulation.25,26 The protocol studied had certain modifications compared to the NPP–FET protocol described by Godinho et al.14 We used intramuscular and vaginal progesterone gel for LPS in all the patients and transferred day 3 or day 5 embryos, whereas Godinho et al. used vaginal micronised progesterone and added intramuscular progesterone every two days in patients where serum progesterone on the day of FET was < 8.5 ng/ml with blastocyst transfer on day 6. To our knowledge, this is the first study to assess ovulation and CL activity in NPP-FET cycles where natural progesterone is used as LPS.
Materials and methods
Study Design
A prospective observational study carried out at a tertiary referral center, Sadbhavna Medical & Heart Institute, Punjab, India from March 2024 to June 2025. Ethics approval was obtained from Institutional Ethics Committee (Reference number: Trg9(310)2024/3561). This study was conducted in strict accordance with the ethical principles outlined in Helsinki Declaration.Written informed consent was taken from all participants.
There were three cohorts of participants
-
NPP-FET cycles (n= 28): This cohort included women undergoing NPP-FET with self or donor oocytes with an ongoing pregnancy (OP) beyond 12 weeks. Inclusion criteria were age between 21 and 40 years, body mass index (BMI) ≤ 35 kg/m2 and regular menstrual cycles (cycle interval 21–35 days).
-
AC-FET cycles (n=11): Patients 21–50 years of age with a BMI ≤ 35 kg/m2, with anovulation, polycystic ovary syndrome or menopause and with an OP beyond 12 weeks were included in this cohort.
Both cohorts of FET cycles had a normal endocrinology work up (thyroid and prolactin), a normal hysteroscopy with no evidence of endometritis on endometrial biopsy. Exclusion criteria included uterine and adnexal anomalies.
- Spontaneously conceived (SC) pregnancies (n=24): Patients between 21–40 years of age with a BMI ≤ 35 kg/m2 and an OP beyond 12 weeks.
Ovarian Stimulation Protocols
Individualized protocols were implemented depending on the age and ovarian reserve in self and donor patients. A gonadotropin releasing hormone (GnRH) antagonist protocol (Ganirelix acetate 0.25 mg, Orgalutran; Organon) starting from day 5/6 with combinations of recombinant follicle stimulating hormone (follitropin beta, Recagon; Organon), recombinant LH (Lutropin alfa, Luveris; Merck) and highly purified human menopausal gonadotropins (Menotropin, Menotas XP; Intas) were administered in doses of 150–300 IU/day. A GnRH agonist (Leuprolide Acetate 4mg, Luprorin; Intas) or recombinant hCG (Choriogonadotropin alfa 250 mcg, Ovitrelle; Merck) was used as the trigger followed by transvaginal oocyte retrieval 36 hours later.
Embryology Lab
Fertilization was through intracytoplasmic sperm injection (ICSI). Embryos were vitrified on day 3 or day 5. Vitrification of embryos was done using the Kitazato media at room temperature. Embryos were placed in the equilibration solution for 12–15 min which was followed by a 90 second exposure to vitrification solution. Thereafter the embryos were loaded on Cryotop device and rapidly cooled by exposing to liquid nitrogen directly. Warming was done by placing the Cryotop to the first warming solution at 37°C. The sample is then transferred through a series of warming solutions with decreasing cryoprotectant concentrations at room temperature. Istanbul Consensus Morphological Assessment was used to grade the embryos.27 The vitrification/thawing protocol utilizes the Cryotop method (Kitazato, Japan) based on the approach described by Kuwayama M.28 In case of day 3 transfer, two or three embryos were transferred and for day 5 transfer, two embryos were transferred.
Endometrial Preparation for FET
-
NPP-FET: A baseline transvaginal ultrasound scan (TVUS) on day 2–5 of menstrual cycle followed by TVUS on day 9/10/11 (2–3 days before anticipated ovulation) was performed. On days 11–14 when EMT was ≥ 7 mm with a trilaminar pattern and serum progesterone was < 1 ng/ml then intramuscular natural progesterone (Susten, Sun Pharma Labs Ltd.) was initiated at a dose of 50 mg on days 1 and 2 followed by daily 100 mg until the day of FET. For day 3 and day 5 embryos, FET was performed on day 4 and 6 of progesterone exposure, respectively. From the evening of the day of FET natural progesterone vaginal gel (Crinone 8%, 90 mg; Merck) was added twice a day till 10–12 weeks. Progesterone injections were continued on alternate days for one week then every third day for another week. This LPS is based on the premise that daily intramuscular progesterone before ET helps achieve adequate serum progesterone levels to promote uterine quiescence.29 Vaginal progesterone gel is subsequently added to reduce the frequency of injections.30 We administer 50 mg for the first two days so as to replicate a gradual physiological increase in serum progesterone levels.
-
AC-FET protocol: From day 1 or 2 endometrial preparation with transdermal estradiol gel (Estrabet gel, 0.06%; Abbott) was initiated at a dose of one application of 2.5 grams gel (equivalent to estradiol 1.5 mg), thrice daily. When EMT was ≥ 7 mm with a trilaminar pattern, progesterone as LPS was initiated similar to that in the NPP-FET cycles and continued till 12 weeks and estradiol gel was tapered by 8 weeks.
Endocrine Monitoring and Assays
In the NPP-FET cohort-Serum progesterone was estimated on day of start of progesterone (P1) and on day of FET (P2) and serum estradiol was determined on day of start of progesterone. Serum relaxin-2 was estimated on day of start of progesterone in the NPP-FET and AC–FET cohorts (R1) and in pregnancies at 6–8 weeks in all the three cohorts (R2).
Hormone assays: Relaxin-2, Progesterone and Estradiol
Serum levels of relaxin-2 were evaluated with an updated and validated enzyme linked immunosorbent assay (ELISA); R&D systems Quantikine ELISA Human Relaxin-2 Immunoassay Minneapolis, Minnesota, USA. The detection range was 7.8–500 pg/ml. The intraassay precision was 3.2% and the intrassay precision was 7.3%. The average % recovery was 101% and specificity with no significant cross–reactivity including relaxin-1 or relaxin-3. Serum samples were collected processed and stored at ≤ –20°C as per the specified instructions. All samples were analysed within one month of collection. Serum progesterone and estradiol estimation was by electrochemiluminesce immunoassay ElecsysIII Cobas e 411 analyzer for progesterone and estradiol respectively (Roche Diagnostics GmbH; Germany) on fresh serum samples. The detection range for progesterone was 0.05–60 ng/ml and for estradiol was 5–3000 pg/ml. The manufacturer’s instructions were followed.
Outcomes
-
The presence of ovulation in NPP-FET cycles-This was indirectly confirmed by the rise in serum relaxin-2 levels in these pregnancies (R2) compared with the levels before starting LPS (R1). Direct confirmation of ovulation by ultrasound documentation of follicular rupture or LH surge monitoring was not performed. As natural progesterone was used as LPS, serum progesterone levels were not estimated for the direct evidence of ovulation.
-
The CL activity in the NPP-FET pregnancies-This was evaluated by comparing the serum relaxin-2 levels (R2) in the NPP-FET cohort to that of the spontaneous conceptions at 6–8 weeks of gestation.
Statistical Analysis
Normality of data distribution was assessed with the Kolmogorov-Smirnov test. Categorical variables were expressed as frequencies and percentages. Results are presented as median with interquartile range (IQR) and compared using the Mann-Whitney U test. R1 and R2 within specific cohorts were compared using the Wilcoxon signed–rank test. Spearman’s correlation coefficient ρ was used to assess the relationship between relaxin-2 levels in early pregnancy and progesterone levels on the day of FET in NPP-FET cycles. A two-sided p value of < 0.05 was considered statistically significant. Data were analyzed using STATA version 16 (StataCorp, Texas, USA).
Results
In the NPP-FET cohort, 34 patients were initially enrolled. Three were excluded due to thin endometrium and three experienced an early miscarriage (n=28). In the AC-FET cohort, 14 patients were enrolled. Two were excluded due to thin endometrium and one patient had an early miscarriage (n=11). The SC cohort included 24 patients who were recruited between 6 and 8 weeks of gestation (n=24). Finally, the results of 63 patients with an OP were analyzed.
The median (IQR) age of patients in the NPP-FET, AC-FET and SC cohorts was 35 (32–38.5), 39 (33–42) and 30 (26–33.5) years respectively. The median age of the NPP-FET cohort was significantly greater than that of the SC cohort but was similar to the AC-FET cohort (p = 0.002 and p = 0.14, respectively). The median (IQR) body mass index (BMI) of the patients in the NPP-FET, AC-FET and SC cohorts was 24.4 (22.1–26.3), 27.4 (26.6–31.1) and 24.2 (23.5–25.7) kg/m2 respectively. The BMI of the NPP-FET cohort was comparable to the AC-FET and SC cohorts (p = 0.06 and p = 0.84, respectively). In the NPP-FET cohort 19 (67.8%) patients used self oocytes and 9 (32.2%) had a donor oocyte cycle. In this cohort 24 (85.7%) patients had a singleton pregnancy and 4 (14.3%) had twins.
The median (IQR) P1 and P2 levels in the NPP-FET cohort were 0.2 (0.1–0.3) ng/ml and 110 (47.3–139.5) ng/ml, respectively. The median (IQR) estradiol levels of the NPP-FET cohort on the day of starting progesterone were 254.4 (140.3–419.0) pg/ml.
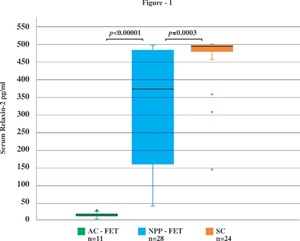
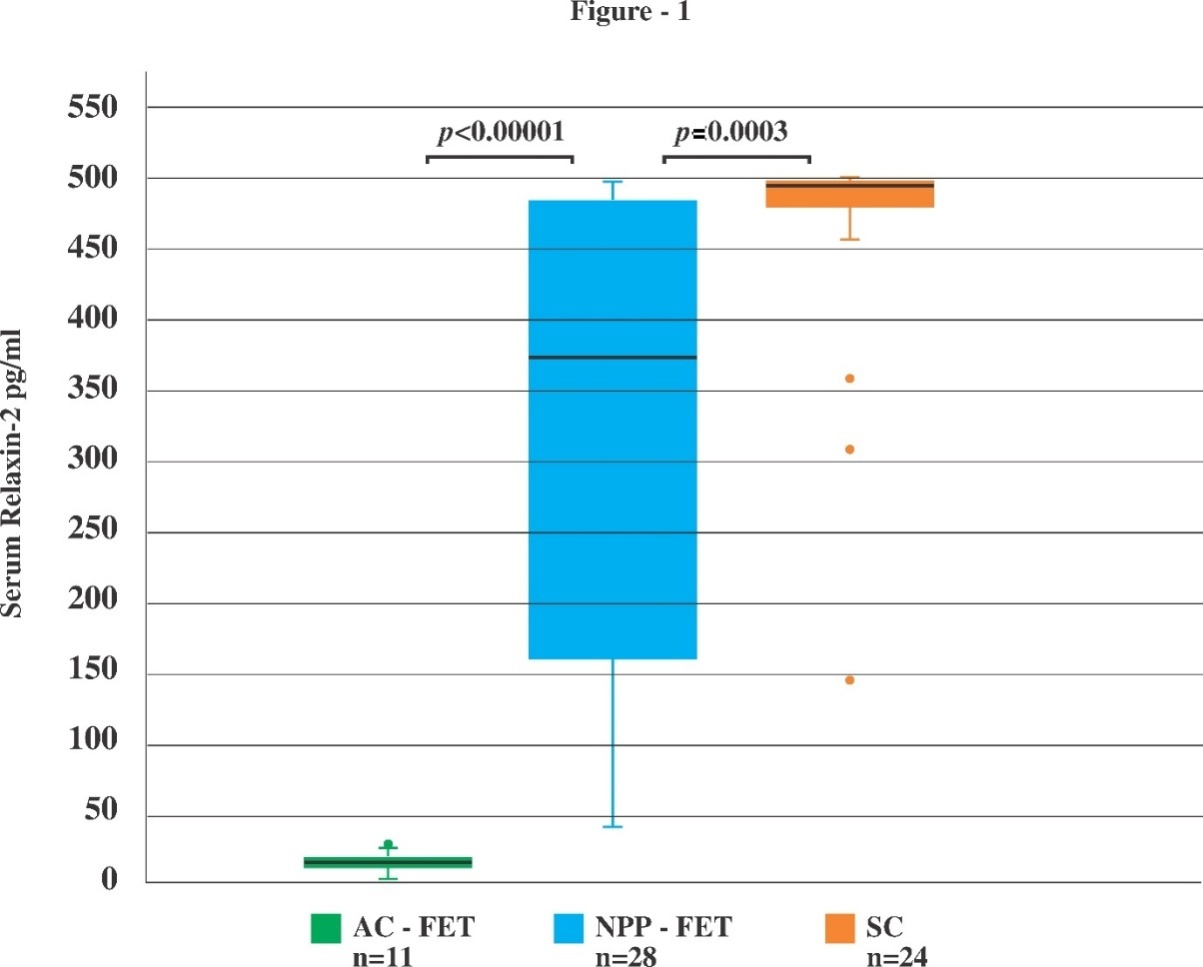
The median (IQR) R1 levels of the NPP-FET and AC-FET cohort were 11.9 (9.3–14.5) pg/ml and 10.3 (7.9–12.5) pg/ml, respectively. There was a significant increase in R2 levels in the NPP-FET cohort compared to the R1 levels, [median (IQR) 368.4 (160–485.2) pg/ml and 11.9 (9.3–14.5) pg/ml, respectively; p < 0.00001]. In 3 of 28 patients of the NPP-FET cohort R2 levels were undetectable or in the lower range of detection. Ovulation was thus indirectly documented in 25/28 (89.3%) of the NPP-FET cycles. As expected, R2 levels in the AC-FET pregnancies were undetectable or in the lower range of detection and were comparable to the R1 levels, [median (IQR) 14.6 (10.3–17.8) pg/ml and 10.3 (7.9–12.5) pg/ml, respectively; p = 0.05] confirming absence of ovulation in the whole cohort.
R2 levels of the NPP-FET cohort were significantly greater than those of the AC-FET cohort [median (IQR) 368.4 (160–485.2) pg/ml and 14.6 (10.3–17.8) pg/ml, respectively; p < 0.00001] and were significantly lower compared to the R2 levels in the SC cohort pregnancies, [median (IQR) 368.4 (160–485.2) pg/ml versus 494.4 (481.6–499.3) pg/ml, respectively; p = 0.0003] as illustrated in Fig1.
There was no significant correlation between R2 and P2 levels in the NPP-FET cohort (ρ = –0.22; p = 0.26).
Discussion
In this study we report indirect evidence for ovulation occurring in 89.3% of the patients with NPP-FET cycles when natural progesterone is used for LPS. The serum relaxin-2 levels in early pregnancy at 6-8 weeks of gestation in the NPP-FET protocol were significantly lower than those in the SC cohort pointing to an altered/inadequate CL activity. To our knowledge this is the first study to report evidence for ovulation and CL activity in NPP-FET cycles with natural progesterone as LPS.
Relaxin-2 is a peptide hormone and is a member of the insulin-like superfamily.There are three known relaxin peptides in humans: relaxin-1, relaxin-2 and relaxin-3 each with varying functions.31 Relaxin-2 is secreted by the CL. Initially in the follicular phase it is either undetectable or in the lower range of detection 10–50 pg/ml, followed by a small peak in the late luteal phase in the ranges reported 30–150 pg/ ml.32,33 In pregnancy it rises, peaks by the end of the first trimester then reduces by 20–50% and remains at intermediate levels thereafter.26,33,34
In IVF pregnancies conceived in the absence of a CL-donor oocyte and AC-FET pregnancies, serum relaxin-2 was either undetectable,25 or in the lower range of detection.35,36 In a study where relaxin-2 was determined in IVF pregnancies without a CL at different time points up to the third trimester, it was consistently undetected.25 Quagliarello et al. confirmed the value of relaxin-2 levels as an index of luteal activity during pregnancy.26 The placenta, decidua also produce relaxin-2 which does not enter the circulation but may have an paracrine or autocrine effect.25,37–39 The median relaxin-2 levels in IVF pregnancies with a single CL as in NC-FET pregnancies and spontaneously conceived pregnancies are reported to be similar, [489.7 versus 517 pg/ml, respectively; p = 0.98] at 11–14 weeks of gestation.36
In our study relaxin-2 levels were either undetectable or in the lower range of detection in the AC-FET cohort pregnancies as also reported in literature.25,35,36 The serum relaxin-2 levels in early pregnancy in the AC-FET cohort were comparable to the relaxin-2 levels prior to pregnancy, p = 0.05 which does not reach the threshold for significance. This is consistent with the expected absence of ovulation and CL activity. In the NPP-FET cohort serum relaxin-2 levels were undetectable or in the lower range of detection prior to starting progesterone as LPS and during pregnancy at 6–8 weeks of gestation there was a significant increase in relaxin-2 levels in 89.3% of patients, an indirect evidence for ovulation in these patients. Absence of ovulation was observed in 3/28 patients. They were given the same LPS and all of them had an uneventful full-term pregnancy with healthy babies. This absence of ovulation can be because of the negative feedback of progesterone on the estradiol induced LH surge as it has a delicate temporal relation to the preovulatory estradiol regarding facilitating or blocking it.20,21,40,41 In this study as all the patients had a proliferative phase of at least 10 days with a minimum EMT of 7 mm and starting natural progesterone at this point of time may not be inhibiting ovulation in most of the patients.
Spontaneous ovulation has been studied in the NPP–FET cycles by two groups with dydrogesterone as LPS. Ovulation was documented in 94.5% of patients by Eggersmann et al.18 and in all patients by Huang et al.16 In the prospective study by Eggersmann et al. with dydrogesterone 30 mg/day as LPS and transfer of day 2–5 embryos, ovulation was evidenced by a serum progesterone level of ≥ 1.5 ng/ml on the day of FET and if negative it was reassessed on the day of pregnancy test. In the retrospective study by Huang et al. with dydrogesterone 40 mg/day as LPS and a single euploid blastocyst transfer, ovulation was documented by ultrasound findings and a rise of serum progesterone > 3 ng/ml within three days of the positive ultrasound findings.
Using natural progesterone as LPS, though may have a different endocrine effect compared to dydrogesterone, CL activity and thus indirect evidenceof ovulation was demonstrated in most of the patients in our study (89.3%).
The median relaxin-2 level in the SC cohort (494.4 pg/ml) was comparable to values reported in literature, including a mean value of 482 pg/ml at 7 weeks of gestation, with levels reported to increase to 517 pg/ml and 542 pg/ml and to range from 1000–2500 pg/ml towards the end of the first trimester in different studies.33,34,36 As our measurements were obtained at 6–8 weeks of gestation, a further rise in relaxin-2 levels would be expected later in the first trimester.
We also evaluated CL activity in pregnancies conceived following the NPP-FET protocol by comparing serum relaxin-2 levels in these pregnancies with those in spontaneously conceived pregnancies at 6–8 weeks of gestation. The median relaxin-2 was significantly lower in the NPP-FET cohort compared to the SC cohort (368.4 pg/ml versus 494.4 pg/ml, respectively; p = 0.0003). This may point to an altered or suboptimal luteal functional activity in the NPP-FET cycles. This may be attributable either to the physiological variation in the normal menstural cycle or to the higher incidence of luteal phase deficiency reported among infertile patients. Though there is no definite biochemical marker for defining an inadequate CL function, progesterone levels less than 10 ng/ml in the mid luteal phase point towards luteal phase defect.42,43 In this study serum progesterone could not be used as a marker of ovulation and CL activity as exogenous natural progesterone was used as LPS. In a recent study 41.5% of the menstural cycles in healthy women had serum progesterone < 30 nmol/L (~9.4 ng/ml) in the mid luteal phase.44 Also serum progesterone levels < 10 ng/ml have been reported in 37% patients one day prior to ET in a NC-FET cycles.45
Further the suboptimal functional activity of the CL observed in the NPP-FET cycles in our study can be related to the endocrine profile of the NPP-FET cycle. Initiating exogenous progesterone prior to ovulation can shorten the proliferative phase compared to what it would have been otherwise, affecting the growth of the dominant follicle and leading to a suboptimal LH surge which may be a causal factor for suboptimal CL function.46
We also observed a weak negative correlation between serum progesterone levels on the day of ET and relaxin-2 levels in early pregnancy in the NPP-FET cohort (ρ = –0.22; p = 0.26). Higher exogenous progesterone levels at the time of FET do not correlate with improved CL function, suggesting that the CL activity observed is independent of the administered exogenous progesterone dose. Furthermore, high serum progesterone levels due to intramuscular administration [median (IQR) 110 (47.3–139.5) ng/ml] may suppress LH pulsatility and potentially attenuate endogenous CL activity during the transition to early pregnancy as suggested in literature.18,47
Strengths and Limitations: This study has several strengths. It was a single-center prospective study with well-defined cohorts. All blood samples were collected and processed within a similar time frame at a single center using validated assays with appropriate quality control metrics. In addition, relaxin-2, a hormone secreted exclusively by the CL during early pregnancy, was used as an indirect marker of ovulation and CL activity providing a physiologically relevant assessment of ovulation in NPP-FET cycles with natural progesterone as LPS.
However, the study also has certain limitations. The relatively small sample size and the absence of a formal a priori power calculation may limit the generalizability of the findings. Ovulation in the NPP-FET cohort was inferred indirectly as direct confirmation through ultrasound documentation of follicular rupture or LH surge monitoring was not performed. Baseline differences in age among the cohorts may have acted as a potential confounding factor in relation to CL activity. Both day 3 or day 5 ET were included with in the same cohort without stratified analysis. Measurement of relaxin-2 levels at later gestational ages, when relaxin-2 levels peak, may have provided additional insight into CL function. The SC cohort values near 500 pg/ml may have been artificially constrained by the upper assay limit. This could mean that the true difference between the NPP-FET and SC cohorts may be larger than reported.
Conclusion
LPS with natural progesterone in NPP-FET cycles preserves spontaneous ovulation in most of the patients. This preliminary data reinforces the reported advantages of the NPP-FET protocol, being simple, preserving the natural proliferative phase, ovulation occurring in most of the patients and ensuring progesterone support from luteal day one. However the CL functional activity may be altered or suboptimal in this protocol. The clinical significance of this observation in relation to further increasing the dose of progesterone or adding another progesterone for LPS and its effect on obstetrical outcomes needs to be explored. Future studies should evaluate the optimal dose, type, and timing of progesterone initiation in the NPP-FET protocol, with systematic evaluation of CL activity and associated clinical outcomes.
Acknowledgements
The authors would like to thank all the patients and health-care providers.
Declaration of Generative AI and AI-assisted Technologies
To prepare this manuscript artificial intelligence (ChatGPT, OpenAI, USA) was used for language refinement and grammar correction during manuscript preparation. All scientific content, interpretation, and final editing were performed by the authors and they take full responsibility for the article content.
Funding Statement
No Financial grants
Conflict of Interest Statement
None of the authors have any conflict of interest
CRediT Authorship Contribution Statement
- Conceptualization: Monica Varma, Preetkanwal Sibia
- Data curation: Monica Varma, Preetkanwal Sibia, Himanshu Bansal, Samman Verma
- Formal analysis: Monica Varma, Sarabpreet Singh, Himanshu Bansal, Samman Verma, Tarundeep Bhatia
- Investigation: Monica Varma
- Methodology: Monica Varma, Sarabpreet Singh
- Software: Samman Verma
- Supervision: Preetkanwal Sibia, Tarundeep Bhatia
- Validation: Sarabpreet Singh, Himanshu Bansal, Samman Verma
- Visualization: Monica Varma
- Writing – original draft: Monica Varma, Sarabpreet Singh
- Writing – review and editing: Monica Varma, Preetkanwal Sibia, Sarabpreet Singh, Himanshu Bansal, Samman Verma, Tarundeep Bhatia
Attestation Statements
-
Data related to any of the subjects in this study has not been published previously.
-
All study and manuscript data will be made available to the journals upon request before and/ or after manuscript publication for review or query.
-
The authors followed the appropriate checklist for this study design (STROBE guidelines).
Data Sharing Statement
Data available upon reasonable request from the corresponding author, Monica Varma, Email: drmonicavarma@yahoo.co.in
Capsule
In natural proliferative phase frozen embryo transfer protocol with natural progesterone as luteal support, ovulation is preserved in 89.3% of patients. Corpus luteal activity is significantly lower compared to spontaneous conceptions.